Microbiological Regulation Consulting
About Microbiological Regulation
Microbiological regulation plays a crucial role in ensuring the safety and quality of pharmaceuticals, food, cosmetics, and medical devices. At Microbiological regulation consulting, we specialize in providing comprehensive guidance on the specific microbiological regulatory requirements for each of these industries. Our consulting services are designed to help businesses navigate the complex landscape of microbiological regulations, ensuring compliance and mitigating risks associated with microbial contamination. Microbiological regulatory requirements for different Industries:
Pharmaceuticals
- Sterility Testing: Pharmaceutical products intended for parenteral administration or sterile applications must undergo sterility testing to ensure the absence of viable microorganisms.
- Microbial Limits: Pharmaceutical products, including non-sterile products, have specific microbial limit requirements to control the level of viable microorganisms.
- Environmental Monitoring: Pharmaceutical manufacturing facilities are required to implement environmental monitoring programs to assess the microbial contamination control measures in place.
Food
- Pathogen Testing: The food industry has stringent regulations regarding the presence of specific pathogens, such as Salmonella, Escherichia coli (E. coli), Listeria monocytogenes, and Campylobacter spp., in food products.
- Total Plate Count: Microbiological testing is conducted to determine the total viable microbial count in food products, which helps assess overall product quality and safety.
- Indicator Organisms: Specific indicator organisms, such as coliforms, are used as indicators of potential fecal contamination and overall hygiene in food processing.
Cosmetics
- Preservative Efficacy Testing: Cosmetics containing water or water-based ingredients are required to undergo preservative efficacy testing to ensure the effectiveness of preservatives in preventing microbial growth.
- Microbial Limits: Regulatory guidelines specify acceptable limits for microorganisms, including total viable count and specific pathogens, in cosmetic products.
- Good Manufacturing Practices (GMP): Cosmetics manufacturers need to adhere to GMP guidelines, which include requirements for facility hygiene, equipment cleaning, and personnel practices to prevent microbial contamination.
Medical Devices
- Bioburden Testing: Medical devices are subjected to bioburden testing to assess the level of microbial contamination on the device before sterilization.
- Sterility Testing: Sterile medical devices must undergo sterility testing to ensure the absence of viable microorganisms after the sterilization process.
- Environmental Monitoring: Manufacturing facilities of medical devices are required to implement environmental monitoring programs to assess and control microbial contamination in the production environment.

Our Microbiological Regulation Consulting Services
Regulatory Compliance Assessment
- Regulatory Landscape Analysis: We conduct an in-depth analysis of the microbiological regulations applicable to your industry and region, identifying the key requirements and guidelines that pertain to microbiology.
- Gap Analysis: Our consultants assess your existing practices and procedures against the regulatory requirements, identifying areas that require attention and improvement to ensure compliance.
Compliance Strategy Development
- Customized Compliance Plans: Based on the regulatory assessment, we develop tailored compliance strategies and action plans to address identified gaps and ensure adherence to regulations.
- Documentation and Standard Operating Procedures (SOPs): Our experts assist in the preparation and review of necessary documentation, including SOPs, testing protocols, and quality assurance procedures, to support compliance efforts.
Microbiological Testing and Analysis
- Method Validation: We guide validating microbiological testing methods according to regulatory guidelines, ensuring their accuracy, reliability, and effectiveness.
- Test Frequency and Sampling Plans: Our consultants recommend appropriate test frequencies and sampling plans that align with regulatory requirements, ensuring comprehensive testing coverage.
- Result Interpretation and Reporting: We assist in interpreting microbiological test results, identifying potential risks or deviations, and generating accurate and comprehensive reports that comply with regulatory standards.
Quality Management Systems
- Good Manufacturing Practices (GMP): We help you establish or enhance GMP-compliant quality management systems, including documentation control, training programs, and change control processes, to ensure microbiological compliance.
- Environmental Monitoring: Our consultants guide implementing effective environmental monitoring programs to assess and control microbial contamination risks within your manufacturing or processing facilities.
Regulatory Updates and Training
- Regulatory Monitoring: We stay up-to-date with the latest developments and revisions in microbiological regulations, keeping you informed of any changes that may impact your operations.
- Training Programs: Our consultants offer customized training programs to educate your staff on microbiological regulations, testing procedures, and best practices, empowering them with the knowledge to comply with regulations effectively.
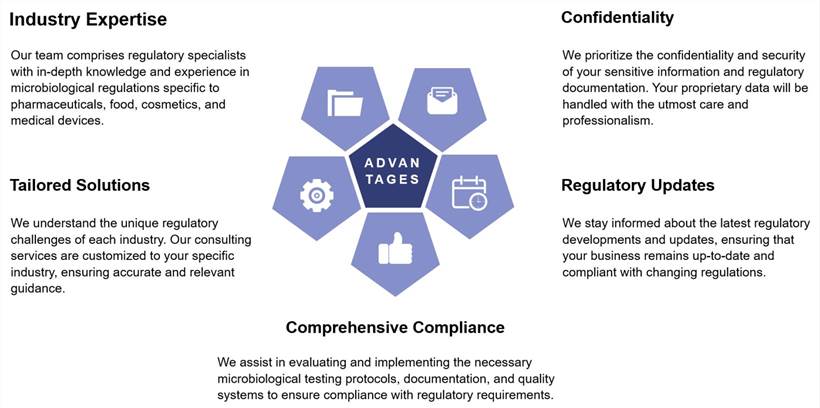
Why Choose Our Microbiological Regulation Consulting?
Contact Us
To ensure compliance with microbiological regulations in pharmaceuticals, food, cosmetics, or medical devices, contact microbiological regulation consulting today. Our team of experts is dedicated to providing tailored guidance and support to meet your specific microbiological regulatory needs.
! For Research Use Only.



